|
A red tomato, for example, is bright red because it reflects red light while absorbing all the other colors of the rainbow. Although colored lights produce their colors, objects are colored because they preferentially reflect a certain color from the white light that shines on them. The color of objects comes from a different mechanism than the colors of neon and other discharge lights. Indeed, the electron configuration of Se is 4 s 23 d 104 p 4, as expected. This means that its electron configuration should end in a p 4 electron configuration. It is in the fourth column of the p block. Consider Se, as shown in Figure 10 “Selenium on the Periodic Table”. Similarity of valence shell electron configuration implies that we can determine the electron configuration of an atom solely by its position on the periodic table. However, many similarities do exist in these blocks, so a similarity in chemical properties is expected. In the d and f blocks, because there are exceptions to the order of filling of subshells with electrons, similar valence shells are not absolute in these blocks. This is strictly true for all elements in the s and p blocks. Elements in each column have the same valence shell electron configurations, and the elements have some similar chemical properties. The same concept applies to the other columns of the periodic table. The organization of electrons in atoms explains not only the shape of the periodic table but also the fact that elements in the same column of the periodic table have similar chemistry. Because much of the chemistry of an element is influenced by valence electrons, we would expect that these elements would have similar chemistry- and they do. However, even these small quantities are required for the body to function properly.Īs previously noted, the periodic table is arranged so that elements with similar chemical behaviors are in the same group.They all have a similar electron configuration in their valence shells: a single s electron. These last three metals are not listed explicitly in Table 2.1.2, so they are present in the body in very small quantities. Cobalt is a necessary component of vitamin B-12, a vital nutrient. 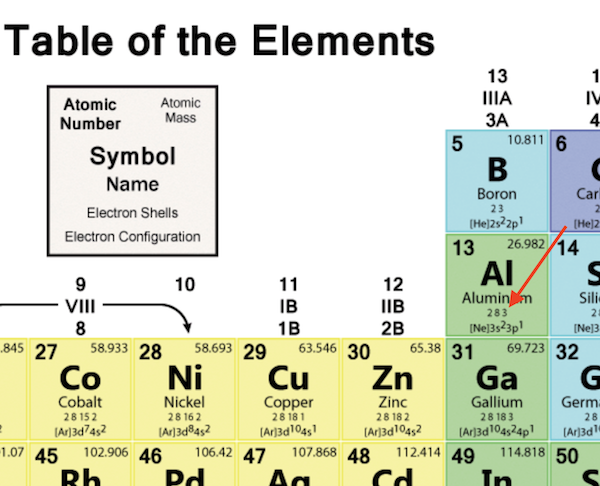
Manganese is needed for the body to metabolize oxygen properly. Copper is also needed for several proteins to function properly in the body. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs.  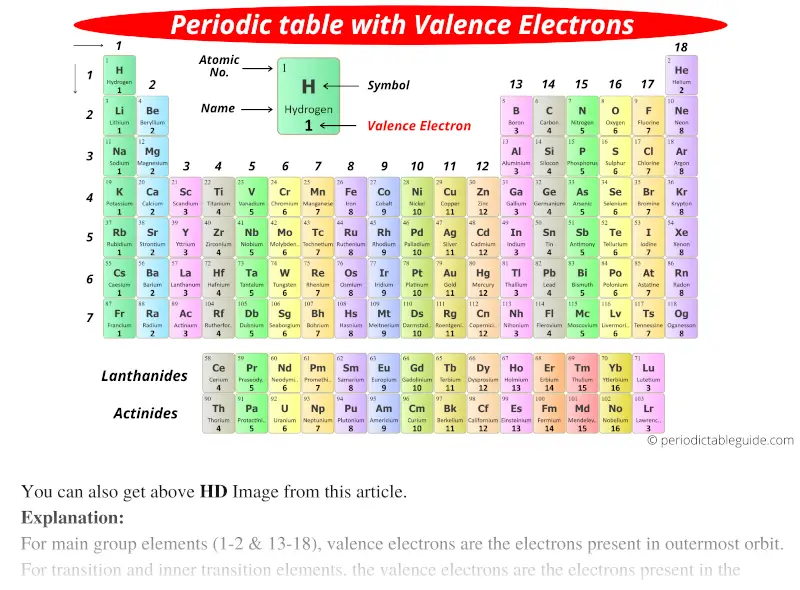
The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
